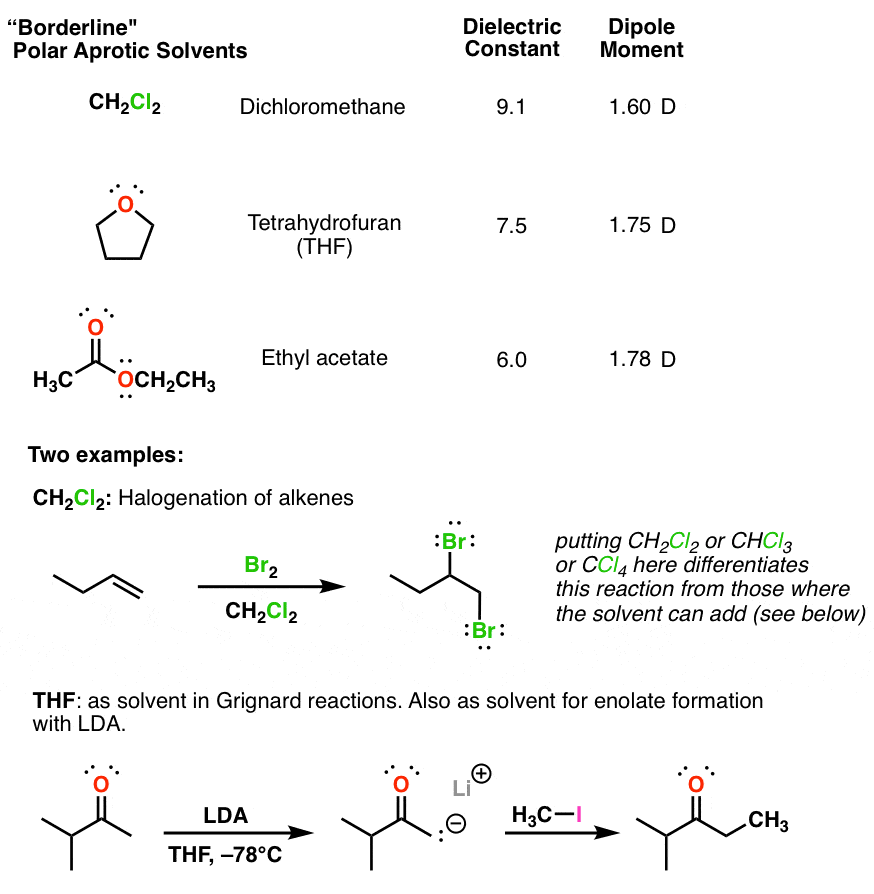
Rationalizing the Mechanism of Peroxyformate Decomposition: Computational Insights To Understand Solvent Influence - Garay‐Ruiz - 2021 - Chemistry – A European Journal - Wiley Online Library

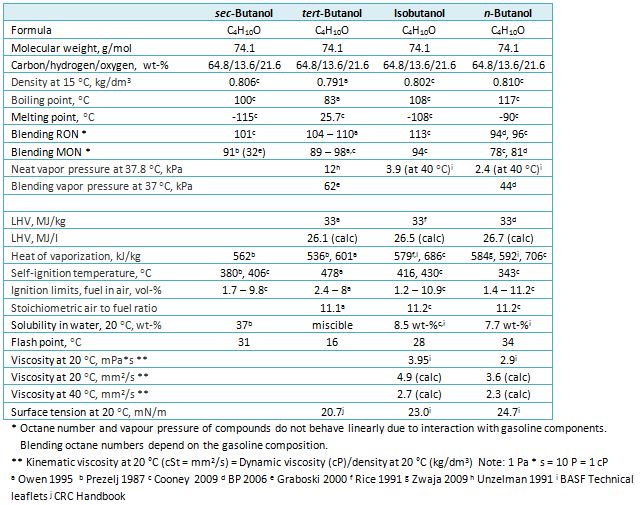
1-Butanol as a Solvent for Efficient Extraction of Polar Compounds from Aqueous Medium: Theoretical and Practical Aspects | The Journal of Physical Chemistry B
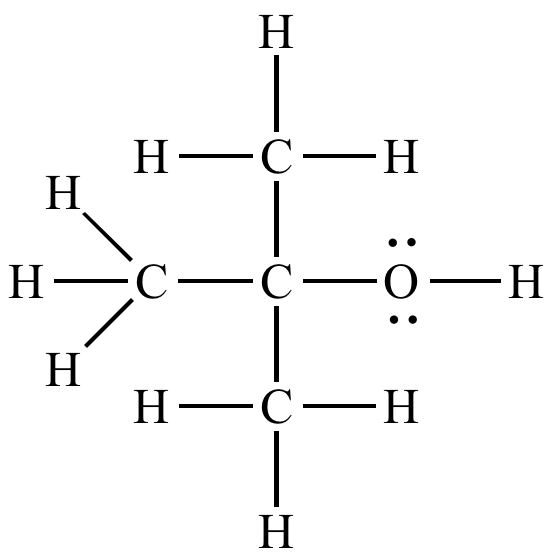
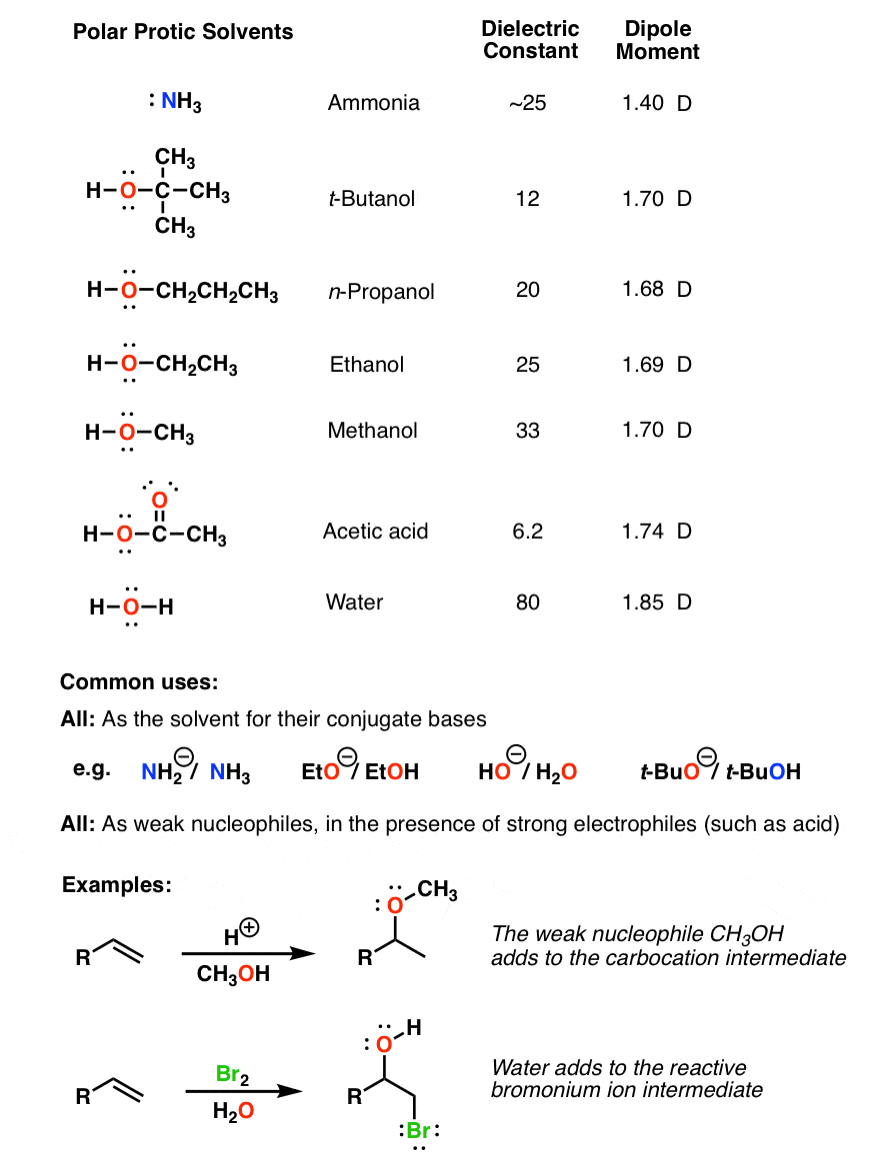
![FL2 C/P #10] How is "2-butanone" more polar than "n-butanol"? I thought presence hydroxyl makes things more polar? : r/Mcat FL2 C/P #10] How is "2-butanone" more polar than "n-butanol"? I thought presence hydroxyl makes things more polar? : r/Mcat](https://i.redd.it/dmcr3fgx2pl11.jpg)