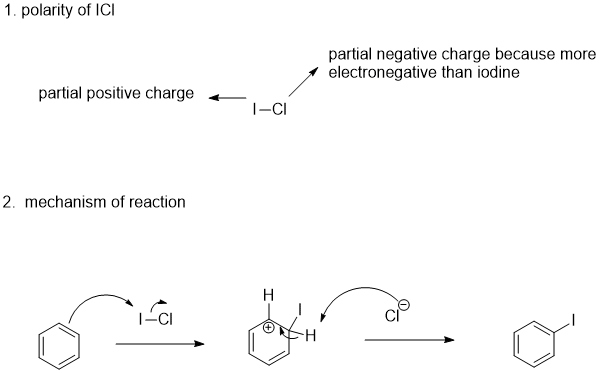
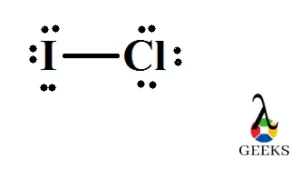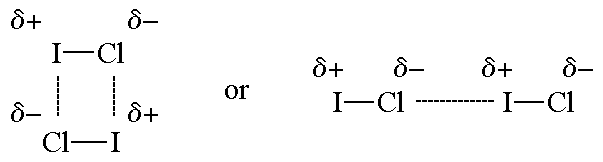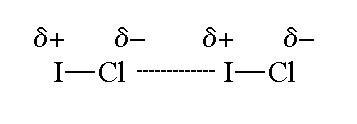SOLVED: Rationalize the difference in boiling points in each pair: a. CHCl3 (61 degrees C) and CHBr3 (150 degrees C) b. Br2 (59 degrees C) and ICl (97 degrees C)

Why is ICl_{2}^{-} a non-polar molecule whereas ICl_{2}^{+} is a polar molecule? | Homework.Study.com

Why is ICl_{2}^{-} a non-polar molecule whereas ICl_{2}^{+} is a polar molecule? | Homework.Study.com